Nanobodies
Nanobodies are a unique kind of monoclonal antibody derived from a camelid IgG variant, consisting of a single heavy-chain variable domain that can bind its antigen as strongly as a standard antibody. As nanobodies lack a light chain, they are both significantly smaller than standard antibodies, and have unique flexibility at their antigen-binding interface. This combination allows nanobodies to bind in different modes than typical antibodies, covering more chemical space and allowing binding to epitopes otherwise inaccessible to antibodies. Nanobodies are also significantly smaller (~15 kDa) and more stable than standard antibodies, and can be easily genetically engineered for additional functionality. With these advantages in mind, we and many others are actively pursuing nanobodies as improved reagents for multiple applications: antibody therapy and diagnostics, affinity isolations, imaging, and more.
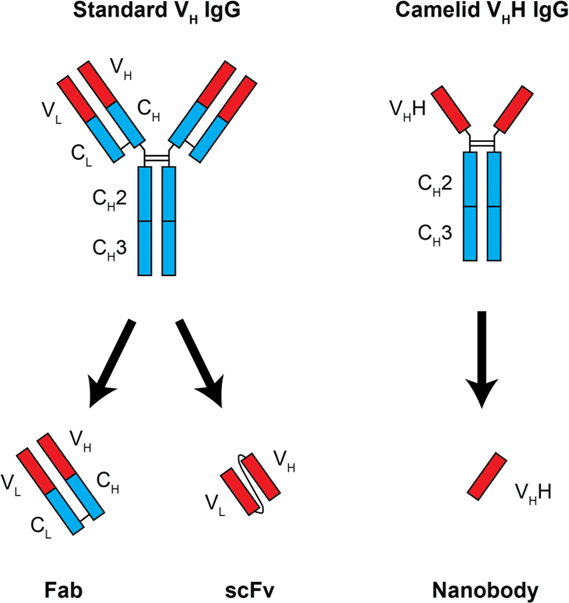
IgG variants and their derivatives. Standard IgG structures contain both heavy and light chains. Variable regions from both chains must be combined for use as an Fab fragment or for recombinant expression as a linked scFv. The camelid V H H IgG variant has only heavy chains. The single variable region can be cloned and expressed independently as a nanobody.
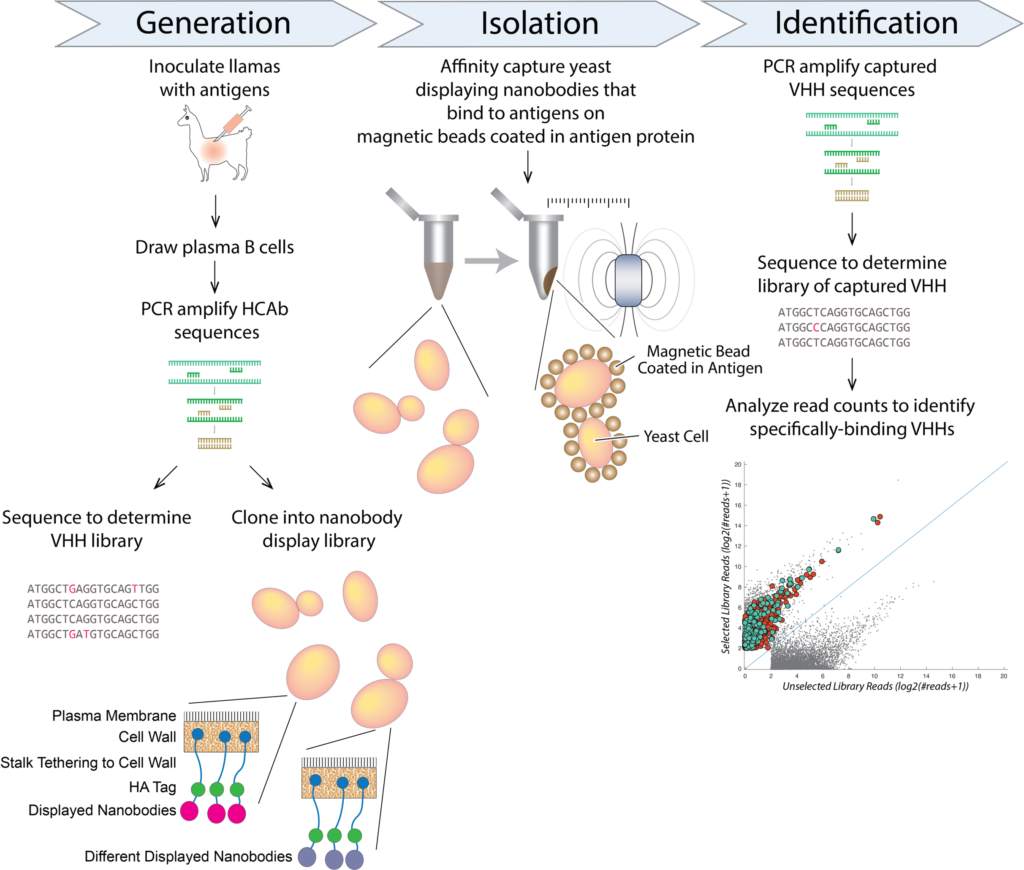
Seq/MS nanobody generation pipeline
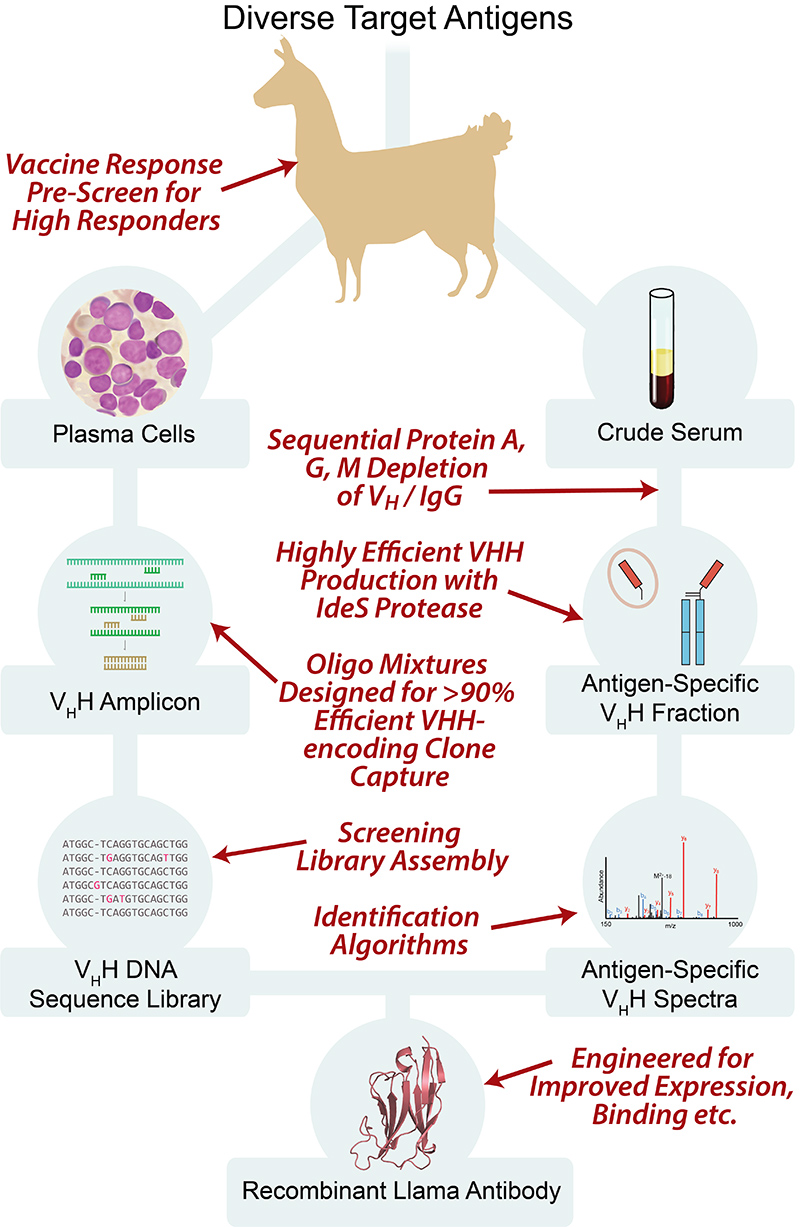
We have developed and optimized a new technology, termed Nanobody Seq/MS, which combines deep sequencing with mass spectrometry to rapidly generate large numbers of bacterially-expressed nanobodies with extremely high affinity and specificity. In our nanobody development pipeline, we first immunize llamas with the antigen target. After a strong immune response is elicited, we collect lymphocytes from bone marrow, highly enriched for active B cells. We then isolate cDNA from these cells, and perform high-throughput sequencing on the PCR-amplified variable region (VHH) of heavy-chain-only IgG variants (HCAbs). In parallel, we collect animal sera, and affinity purify HCAbs with strong affinity and specificity to each antigen. The purified HCAbs are then analyzed by MS, and by correlating peptides to the DNA database of full-length sequences, we can identify candidate nanobody clones. We also now utilize a newly-modified orthogonal and complementary approach, based on yeast surface display, to expand our nanobody repertoires.
We first demonstrated the effectiveness of this approach by generating more than 30 high affinity nanobodies against GFP and mCherry.
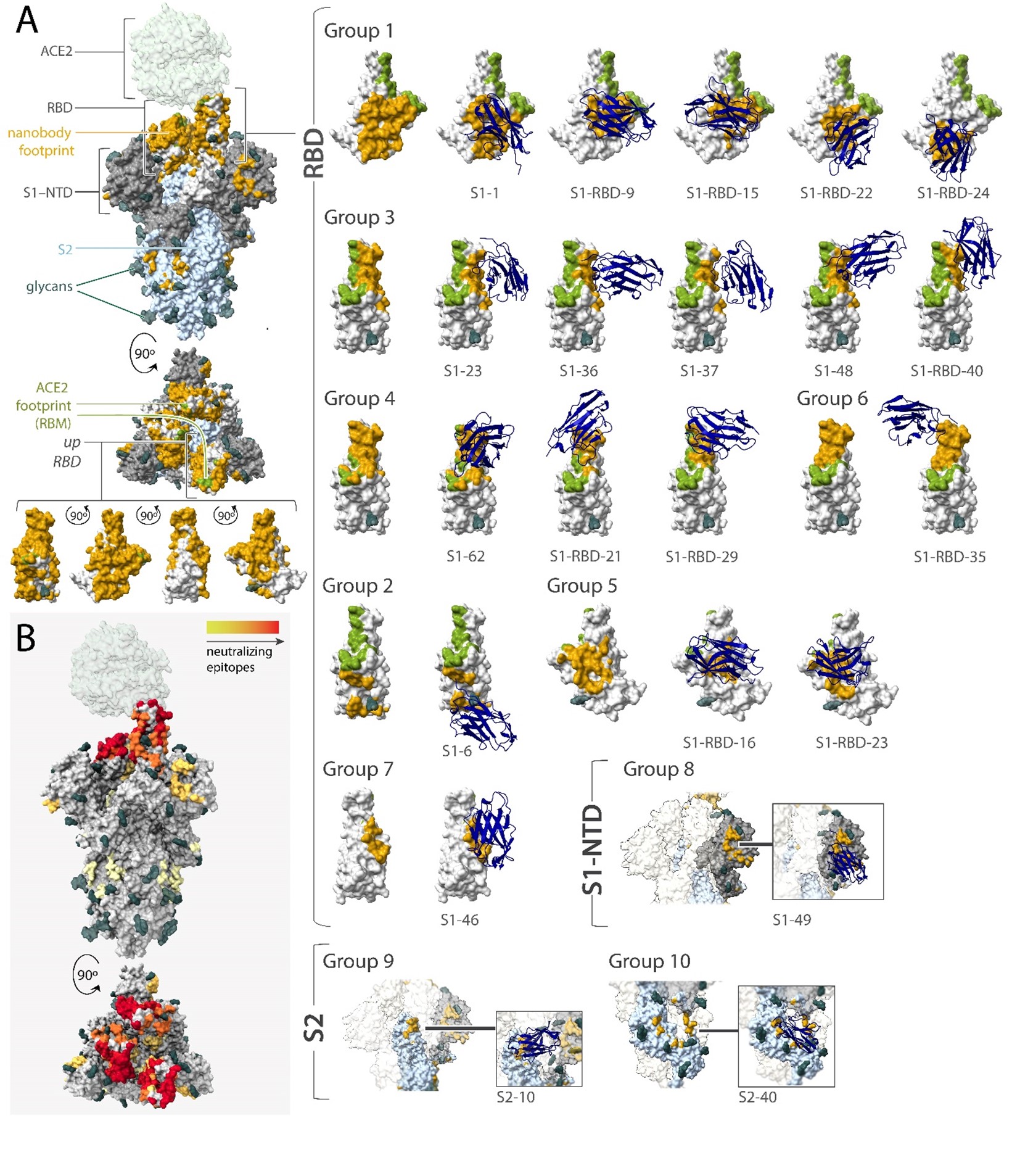
Since this initial proof of principle, we have applied our nanobody generation pipeline to over 30 new antigens, generating a total of more than 500 unique nanobodies. Most recently, we generated a panel of 116 nanobodies against the SARS-CoV-2 spike protein, most of which were capable of neutralizing viral infection. These neutralizing nanobodies act through a diverse range of epitopes and mechanisms, retain activity against the most recent SARS-CoV-2 variants, and can act in synergistic combinations that dramatically increase potency.
Among our current interests, we are developing new nanobodies for use as therapeutics against cancer targets and infectious agents.